1. The development of new and innovative treatment methods aimed at remyelination.
How can we prevent or halt the process of myelin loss and promote the formation of new myelin?
By studying these processes in detail at the cellular and molecular level, we hope to identify potential targets for new treatment methods.
Questions we aim to answer in this research line include:
Which cells present in MS lesions hinder the remyelination process, and how can we create an optimal cellular environment to support the repair process in MS?
Which molecules in MS lesions inhibit remyelination in the affected areas, and how can these inhibitory processes be stopped?
These research questions are focused on reactivating remyelination in affected areas and halting disease progression.
Projects
What goes wrong during the development of MS lesions?
RESEARCHERS – Eva Geerts, Amber Woudstra, Susanne Kooistra, Wia Baron, Bart Eggen
PROJECT: In people with Multiple sclerosis (MS), the myelin sheath—the protective layer surrounding nerve fibers—becomes damaged. Eventually, this layer disappears completely. The gradual deterioration experienced by people with MS is partly caused by the failure of myelin to (fully) repair. In previous research, different types of affected areas (lesions) were mapped in detail, resulting in a model that can predict lesion development. Based on this model, we believe that disrupted cellular processes at the rim of the lesion cause the lesion to slowly expand. At the same time, processes occurring in the center of the lesion appear to hinder myelin repair. Several brain cell types are involved in the clearance of damaged myelin and the subsequent repair process. This project consists of two research lines, in which we investigate both the role of astrocytes (Research Line 1) and
To better understand the characteristics of astrocytes and microglia and their interactions, we use state-of-the-art technology to analyze, cell by cell, what is happening in both the rim and the center of lesions. Subsequently, induced pluripotent stem (
The ultimate aim of this research is to unravel and understand the processes that drive and regulate lesion growth. In addition, we investigate the mechanisms responsible for the transition from a lesion that is still repairable to one that is no longer capable of repair. This research may lead to new therapeutic targets for potential drugs that, in combination with current anti-inflammatory treatments, could truly slow the progression of MS.
Financial contribution from Stichting MS Research (23-733e) (MSCNN program grant)
Astrocytes as novel therapeutic targets for MS
RESEARCHERS – Tamara Hageman, Naomi Dijksman, Susanne Kooistra, Bart Eggen, Wia Baron
PROJECT: Multiple sclerosis (MS) is characterized by the breakdown of myelin. This damaged myelin is usually not repaired, ultimately resulting in the loss of nerve cells.
Recent research findings indicate that this failure of repair is not due to dysfunction of the myelin-producing cells themselves—the oligodendrocytes—but rather to surrounding cells that incorrectly regulate these myelin-producing cells. These surrounding cells, called astrocytes, were long considered passive “bystanders” rather than contributors to the failure of myelin repair. Now that it has become clear that astrocytes are involved in directing myelin repair, it is essential to further investigate the role of these cells and everything they produce.
We aim to answer the following questions: Do astrocytes from people with MS respond differently to inflammation and myelin damage than astrocytes from people without MS—for example, those of their unaffected siblings? Are astrocytes from people with relapsing MS different from those with progressive MS? And do these differences influence the process of myelin repair?
In this study, we use induced pluripotent stem (
The ultimate goal of our research is to identify points of intervention—so-called targets—that would allow us to selectively convert an MS-associated astrocyte into one that promotes myelin repair. This could serve as a starting point for developing a therapeutic strategy aimed at restoring myelin in people with MS.
The ultimate outcome: halting the progression of MS.
Financial contribution from Nationaal MS Fonds

Altered gene activity in MS
Are these genes and changes relevant, and can they be used as targets for therapy?
RESEARCHERS – Janneke Bosma, Bart Eggen, Susanne Kooistra.
PROJECT:
Multiple sclerosis (MS) is a chronic disease of the central nervous system characterized by inflammatory responses, in which myelin is broken down, nerve cells are lost, and scar tissue remains. A better understanding of the disease mechanisms—particularly in relapsing-remitting MS—has led to current therapeutic options, which primarily focus on suppressing inflammatory responses to limit myelin damage. Unfortunately, there is still no effective therapy for progressive MS.
Research questions relevant to addressing this unmet need include: How can we prevent the formation of new lesions? Can we repair existing lesions by stimulating myelin production? Answering these questions may lead to therapeutic targets that can slow MS progression and improve the quality of life of affected individuals.
In previous research, we mapped differences in gene activity between tissues and cells from people with and without MS. This resulted in several extensive datasets identifying genes with altered activity in MS. These genes may represent potential therapeutic targets. However, how these changes in gene activity affect central nervous system tissue and how they contribute to MS pathology remains unclear. Follow-up research, using a novel scientific technique that allows us to map specific cells and precise locations within an MS lesion in great detail, may provide further insight.
In addition, we use human stem cells to study the consequences of altered gene expression on cell–cell interactions and on myelin.
Our ultimate goal is to define a limited number of strong candidate targets which, based on functional analyses, may serve as potential targets for the development of therapies that are effective in progressive MS.
Financial contribution from Stichting MS Research (22-1177)
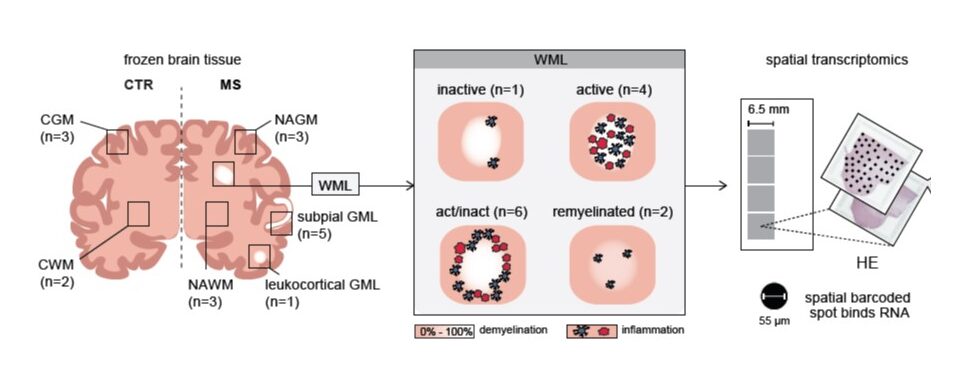
Cel-cel interactions in MS lesions
RESEARCHERS – Janssen Kotah, Mirjam Koster, Susanne Kooistra en Bart Eggen.
PROJECT: Multiple sclerosis (MS) is characterized by the breakdown of myelin, the insulating layer surrounding nerves, for reasons that remain unknown. As a result, so-called lesions develop. Damaged myelin can repair, but sometimes—particularly in progressive MS—repair fails to occur and nerve cells are lost. Crucial questions are: Why and where do lesions arise? Why does myelin repair occur in some cases but not in others?
Using the latest technologies, individual cells within MS lesions can be mapped in detail. This allows us to better understand the changes that occur in individual cells during lesion formation and repair. However, information about where exactly these cells were located within the tissue is lost in this approach. As a result, information about cell–cell contacts—and whether these interactions are disrupted in an MS lesion—is also lost.
In this project, we combine multiple techniques that enable us to map altered cell–cell interactions during the formation and progression of MS lesions.
We then investigate the consequences of these disrupted cell–cell interactions for the functioning of surrounding brain cells using
This research will provide deeper insight into how different brain cell types are involved in the formation and progression of MS lesions. Such insight is essential for identifying new targets to improve treatment strategies for MS.
Financial contribution from Stichting MS Research (20-1104) and a Fund Girn travel grant (
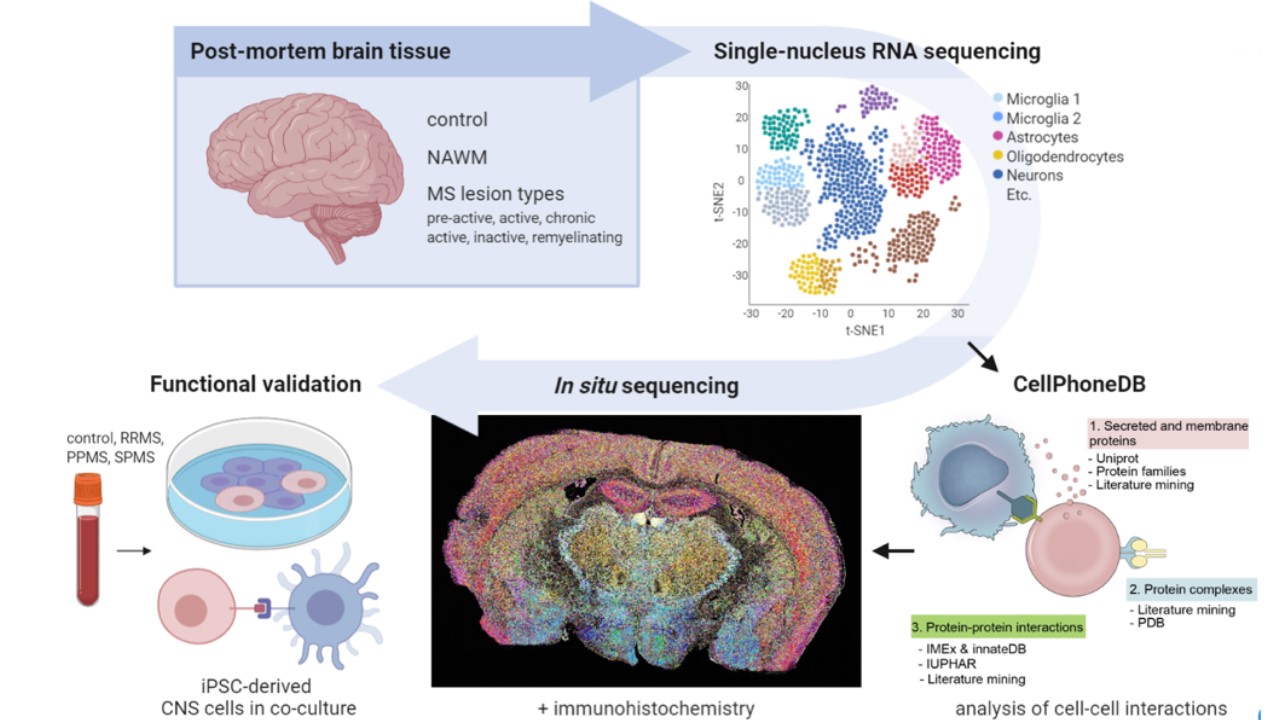
Cellular heterogeneity and cell–cell interactions in the development and progression of MS lesions
RESEARCHERS – Mirjam Koster, Evelyn Wesseling, Susanne Kooistra, Bart Eggen en Wia Baron
PROJECT:
Multiple sclerosis (MS) is a complex disease in which pathology varies considerably between patients. In particular, disease course, the location of lesions within the central nervous system (CNS), and the cell types involved are major sources of variation. Both CNS-resident cell types—including neurons, astrocytes, oligodendrocytes, and
Recent research has demonstrated the presence of cellular heterogeneity within MS lesions; that is, the composition of distinct cell populations within major cell types differs between lesions. At present, little is known about how this cellular heterogeneity contributes to MS pathology and disease progression. This research project aims to understand the role of cellular heterogeneity and cell–cell interactions in the development and progression of MS lesions.
Financial contribution from a GSMS PhD fellowship and from Stichting MS Research via a Fund Girn travel grant (
Immune memory of microglia
RESEARCHERS – Tiago Medeiros Furquim Mendonça, Amalia Dolga, Erik Boddeke, Susanne Kooistra en Bart Eggen
PROJECT: In addition to adaptive immune cells, innate immune cells can also develop immunological memory. The innate immune cells of the central nervous system,
Multiple sclerosis (MS) is an inflammatory, neurodegenerative disease characterized by demyelinated lesions with oligodendrocyte loss and progressive axonal degeneration in the central nervous system (CNS). Microglia, the macrophages of the CNS, are also widely involved in MS. Their role in MS is complex: microglia can have both tissue-supportive and detrimental functions, depending on the stage of MS. How the innate immune system within microglia contributes to MS pathology is the primary focus of this project.
Financial support is provided through a GSMS PhD fellowship and additional funding from Stichting MS Research via a Young Talent Award (22-1149).